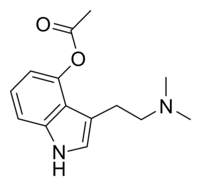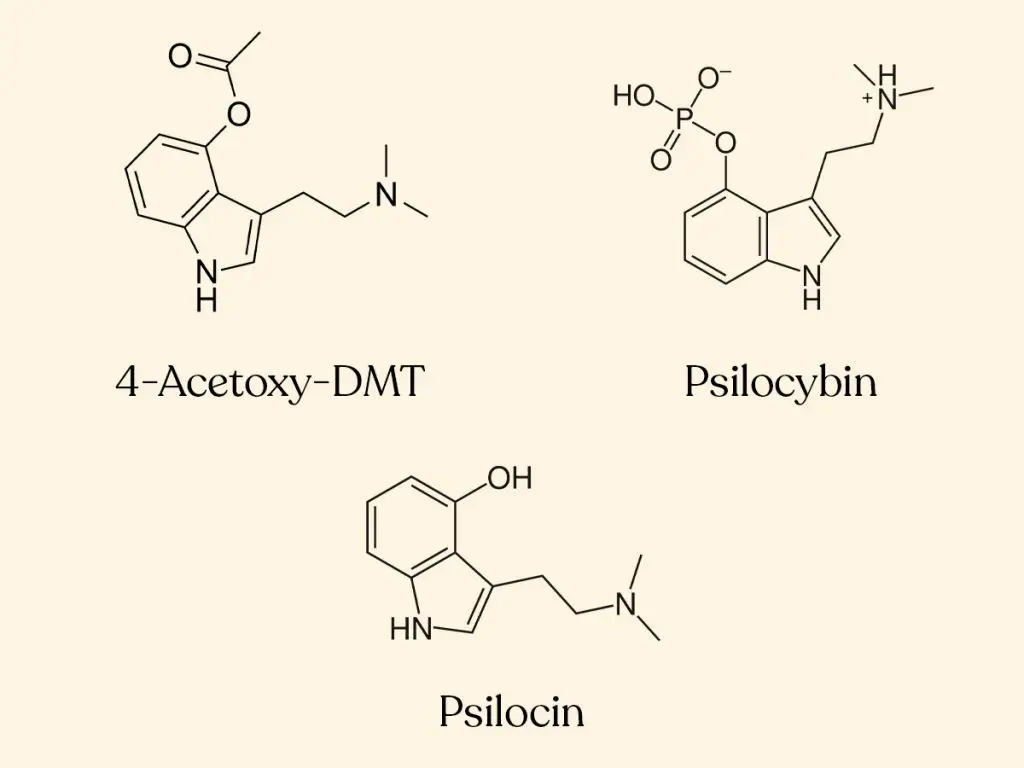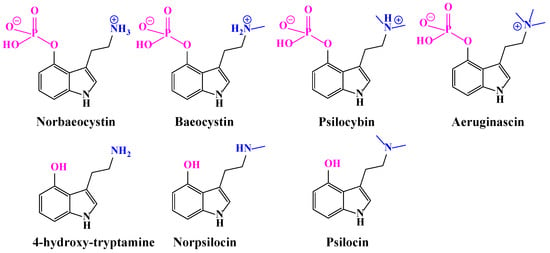
Pharmaceuticals | Free Full-Text | A Review of Synthetic Access to Therapeutic Compounds Extracted from Psilocybe

Psilocin – The “real deal” or an extraction byproduct - Gutman - Journal of Forensic Sciences - Wiley Online Library

Synthesis, Structural Characterization, and Pharmacological Activity of Novel Quaternary Salts of 4-Substituted Tryptamines | ACS Omega

Active Metabolite of Aeruginascin (4-Hydroxy-N,N,N-trimethyltryptamine): Synthesis, Structure, and Serotonergic Binding Affinity. - Abstract - Europe PMC

Structure–Activity Relationships for Psilocybin, Baeocystin, Aeruginascin, and Related Analogues to Produce Pharmacological Effects in Mice | ACS Pharmacology & Translational Science
Concise Synthesis of N,N-Dimethyltryptamine and 5-Methoxy-N,N-dimethyltryptamine Starting with Bufotenine from Brazilian Anadena
Molecules | Free Full-Text | A Comprehensive Analysis of the Metal–Nitrile Bonding in an Organo-Diiron System

Active Metabolite of Aeruginascin (4-Hydroxy-N,N,N-trimethyltryptamine): Synthesis, Structure, and Serotonergic Binding Affinity | ACS Omega
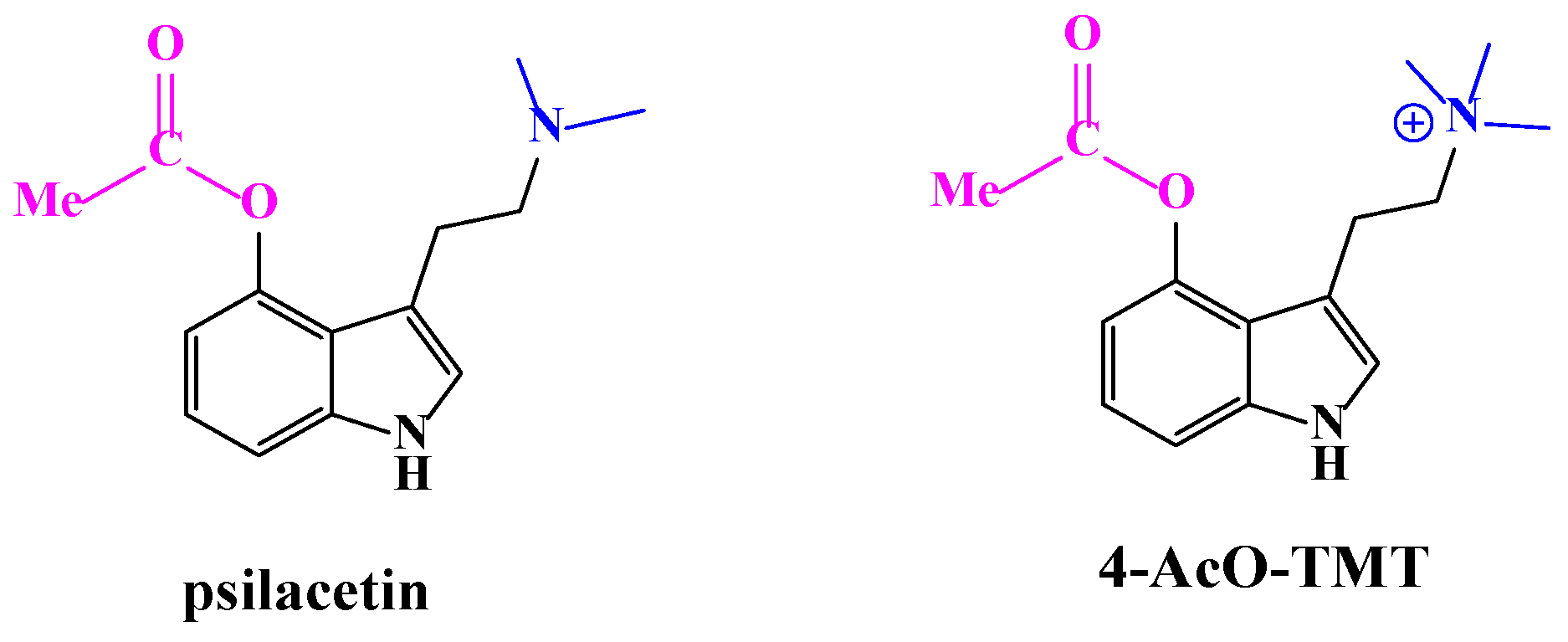
Pharmaceuticals | Free Full-Text | A Review of Synthetic Access to Therapeutic Compounds Extracted from Psilocybe



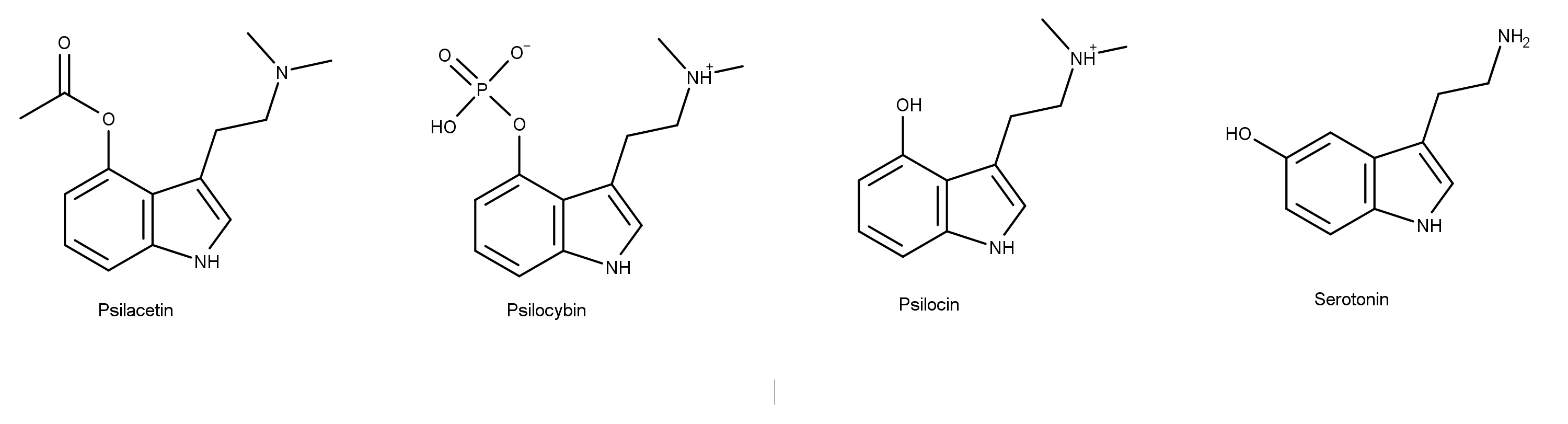
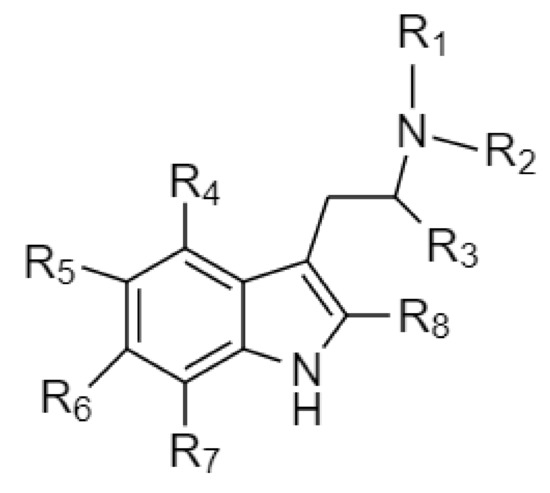

![Synthesis of Psilocybin & Psilocin - [www.rhodium.ws] Synthesis of Psilocybin & Psilocin - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/pictures/psiloscheme1.gif)