Selection of stainless steels for handling sulphuric acid (H2SO4) – British Stainless Steel Association
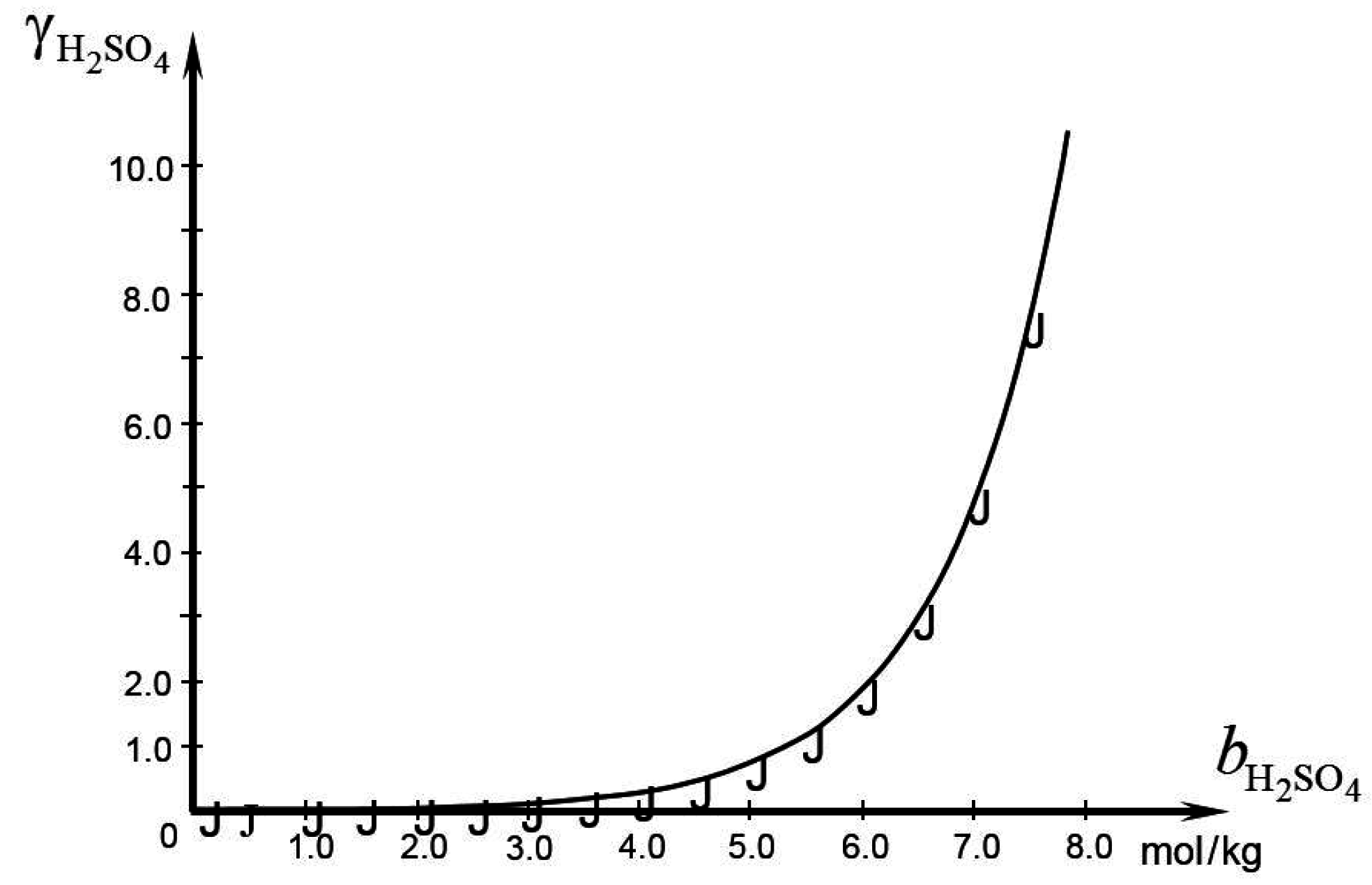
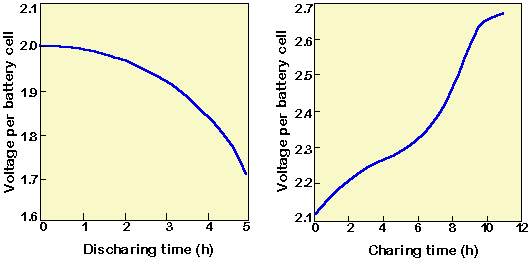
Batteries | Free Full-Text | Electrolyte Additive Concentration for Maximum Energy Storage in Lead-Acid Batteries

Question Video: Determining Which Lead–Acid Batteries Need to Be Recharged Using Relative Density Values for Sulfuric Acid | Nagwa

SOLVED: Battery acid, the electrolyte in all lead acid cell batteries; is sulfuric acid (HzSO4) at a concentration of between [5% and 35%. At this concentration, sulfuric acid is very dangerous and

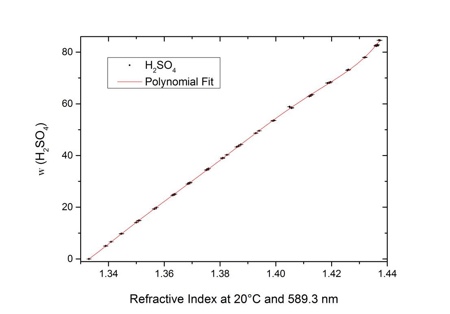
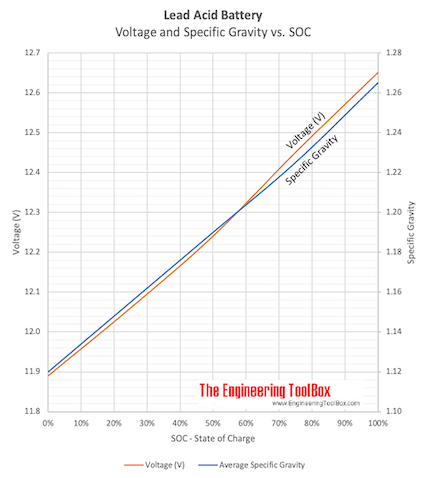
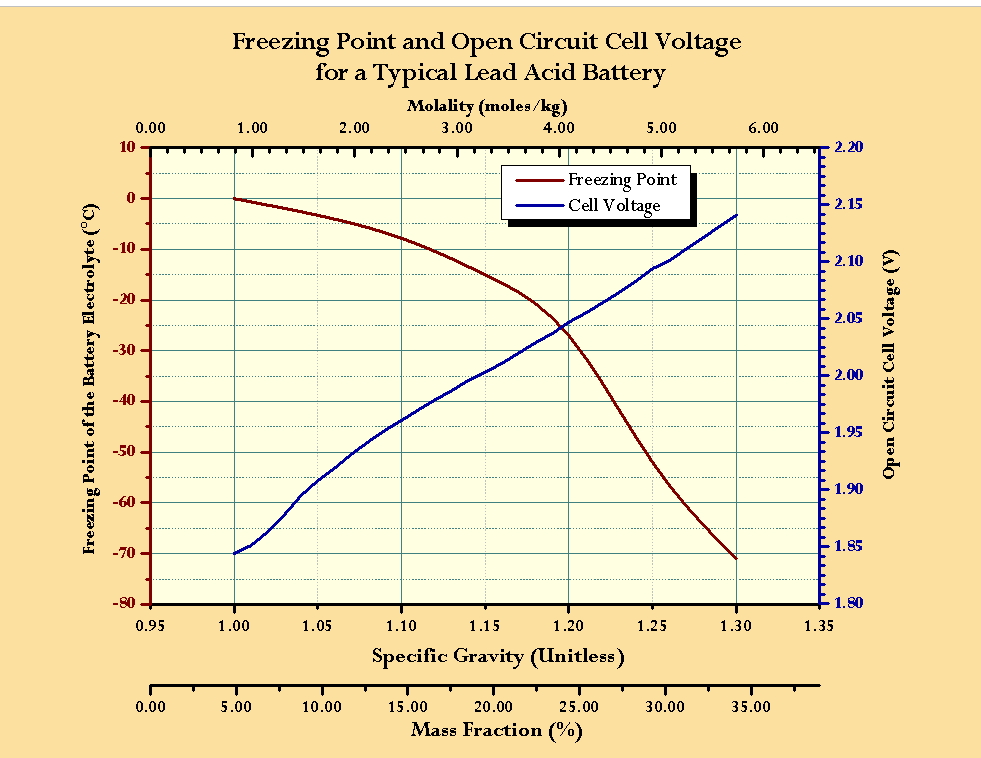
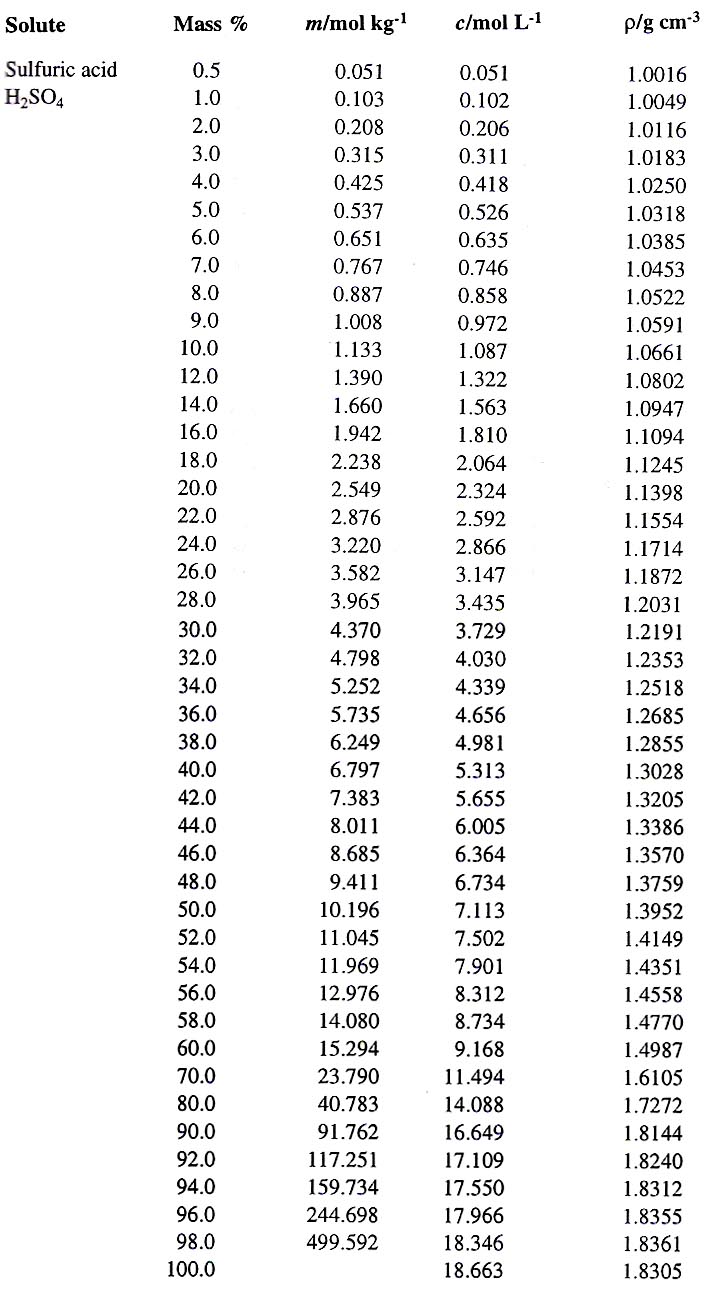
Measuring the density and specific gravity of battery acid in lead acid batteries :: Anton Paar Wiki
How to increase the Sulfuric acid concentration in battery acid which usually contains 35% sulfuric acid and 65% water - Quora

Investigation of the electrochemical behaviour of lead dioxide in aqueous sulfuric acid solutions by using the in situ EQCM technique | SpringerLink

Influence of H2SO4 concentration on lead-acid battery performance: H-type and P-type batteries - ScienceDirect
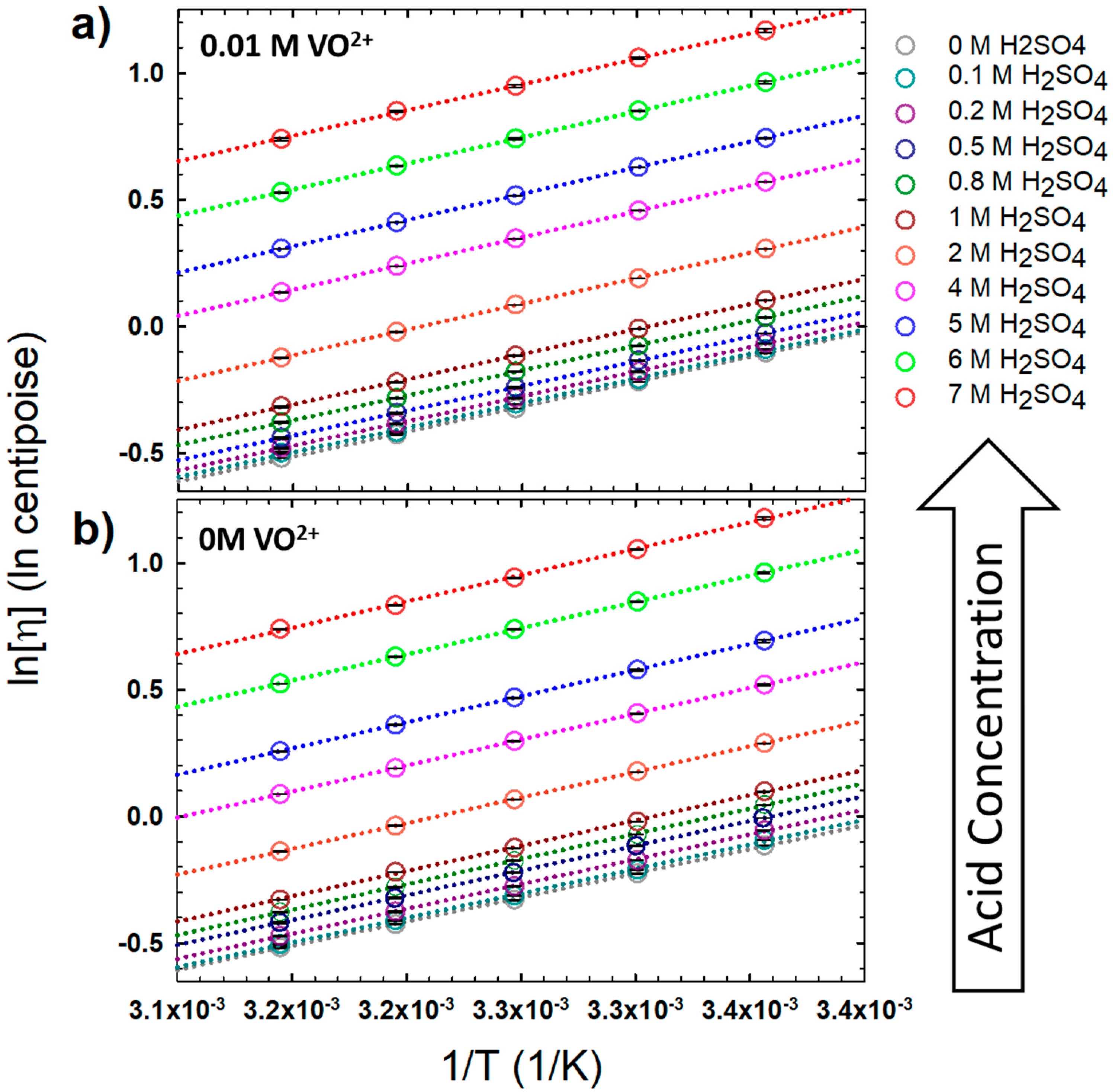
Batteries | Free Full-Text | The Effect of Sulfuric Acid Concentration on the Physical and Electrochemical Properties of Vanadyl Solutions



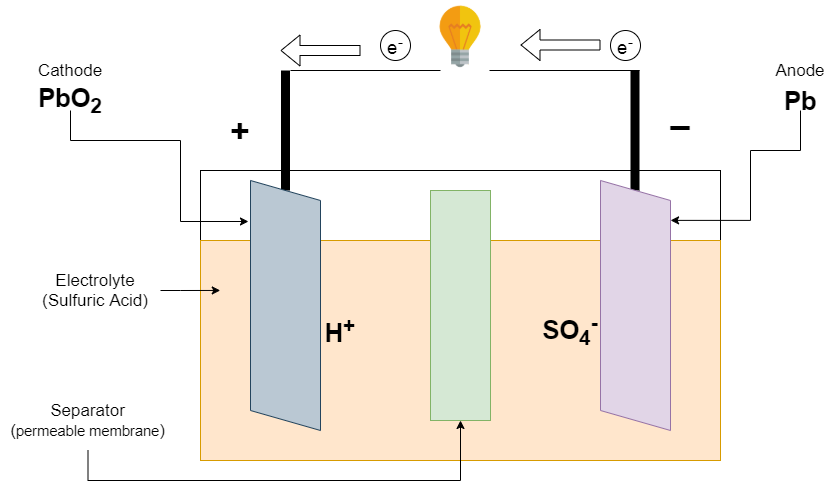

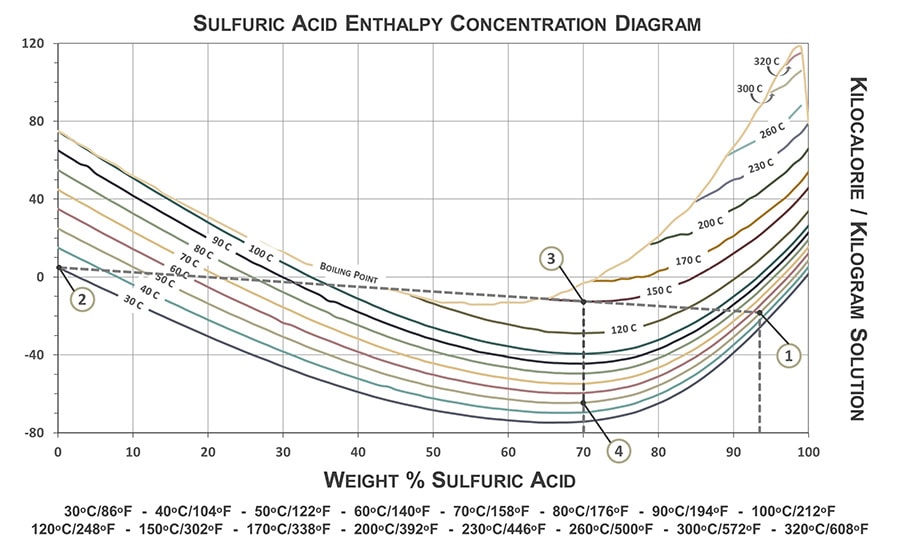
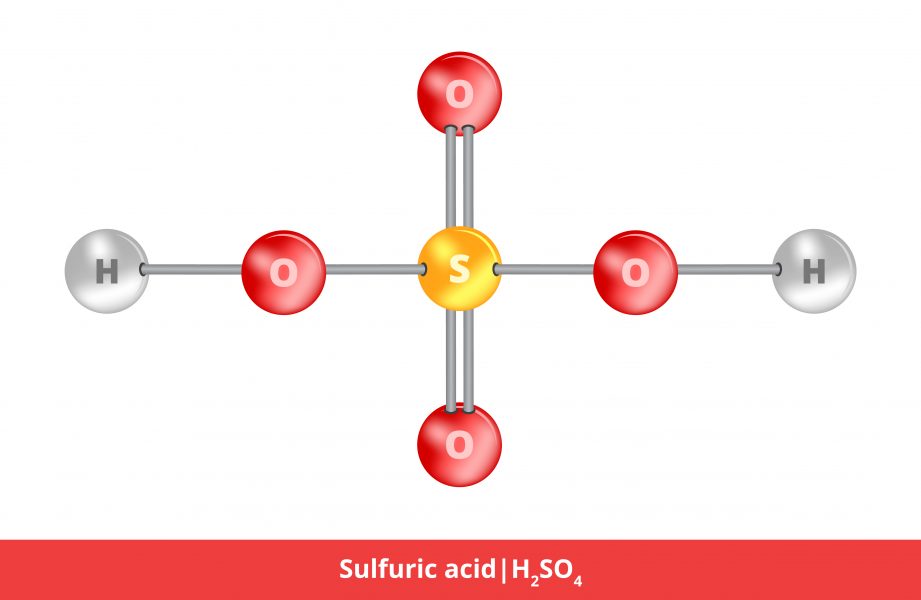
:max_bytes(150000):strip_icc()/car-battery-recycling-container-with-warning-notices-battery-acid-flusco-household-waste-recycling-centre-cumbria-uk-121814398-57a4e5055f9b58974a7355d8.jpg)