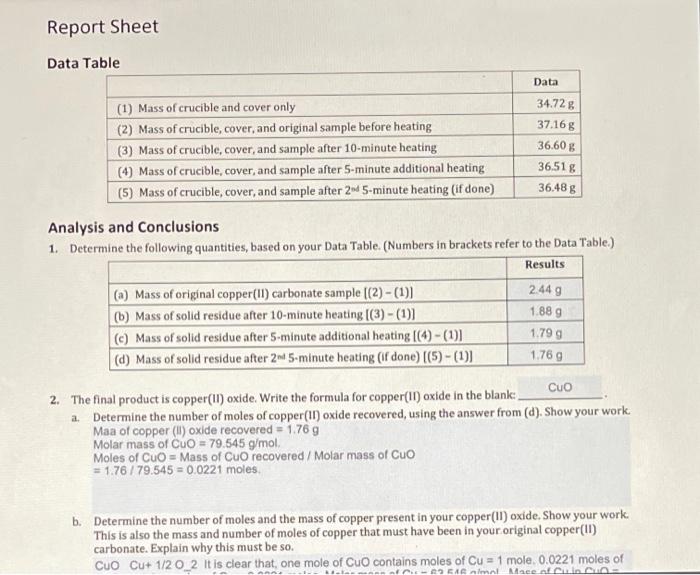
SOLVED: composition OF COPPER CARBONATE Pre-Lab Assigrment Determine the theoretical %Cu in Cuco;, CuCo-H,0 Cus(OH)CO,, Cus(OH)(CO )-H,o NazCu(cOs)z: Put setups and results for all but the first formula Sneen part A of

Reactions of Group 2 Oxides, Hydroxides & Carbonates (2.2.2) | CIE A Level Chemistry Revision Notes 2022 | Save My Exams

Use of Vanadium(V) Oxide as a Catalyst for CO2 Hydration in Potassium Carbonate Systems | Industrial & Engineering Chemistry Research

SOLVED: Q2 Calculate the average atomic mass of magnesium using the following data for three magnesium isotopes. You must show all your work to get credit Isetope IaSS (W)) relativg abundance Mg-24

Metal carbonates are known to undergo thermal decomposition, producing the metal oxide and releasing carbon dioxide. The process is described by the following generic equation, in which M represents an unknown divalent

Calcium carbonate decomposes, on heating , to form calcium oxide and carbon dioxide. When 10 g of - YouTube
Iron is produced by the reduction of iron (III) oxide using carbon monoxide. Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g). How much Fe is produced from 1 kg of Fe2O3? - Quora

gcse Formulae oxides, hydroxides, carbonates, chlorides, sulphates/sulfates, nitrates, salts solubility curves & water of crystallisation data tables KS3 KS4 Science IGCSE chemistry revision notes
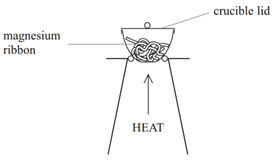
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry