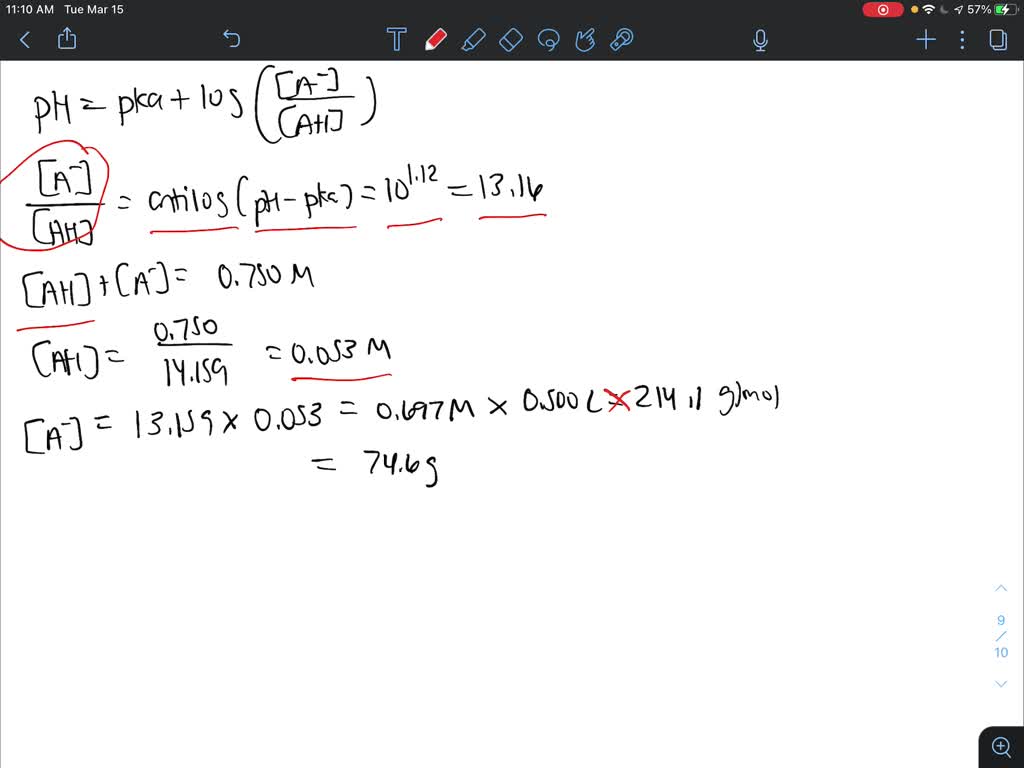
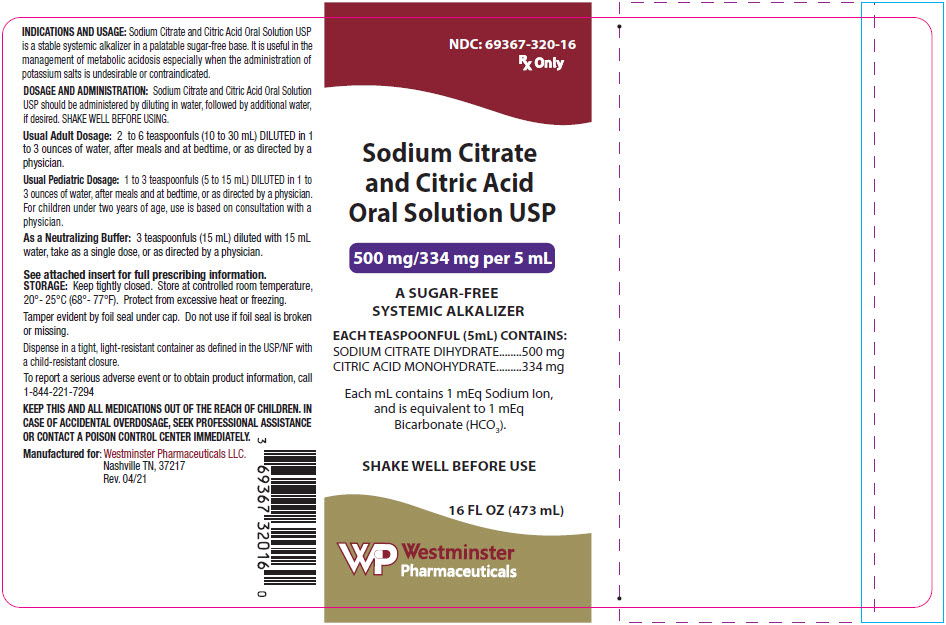
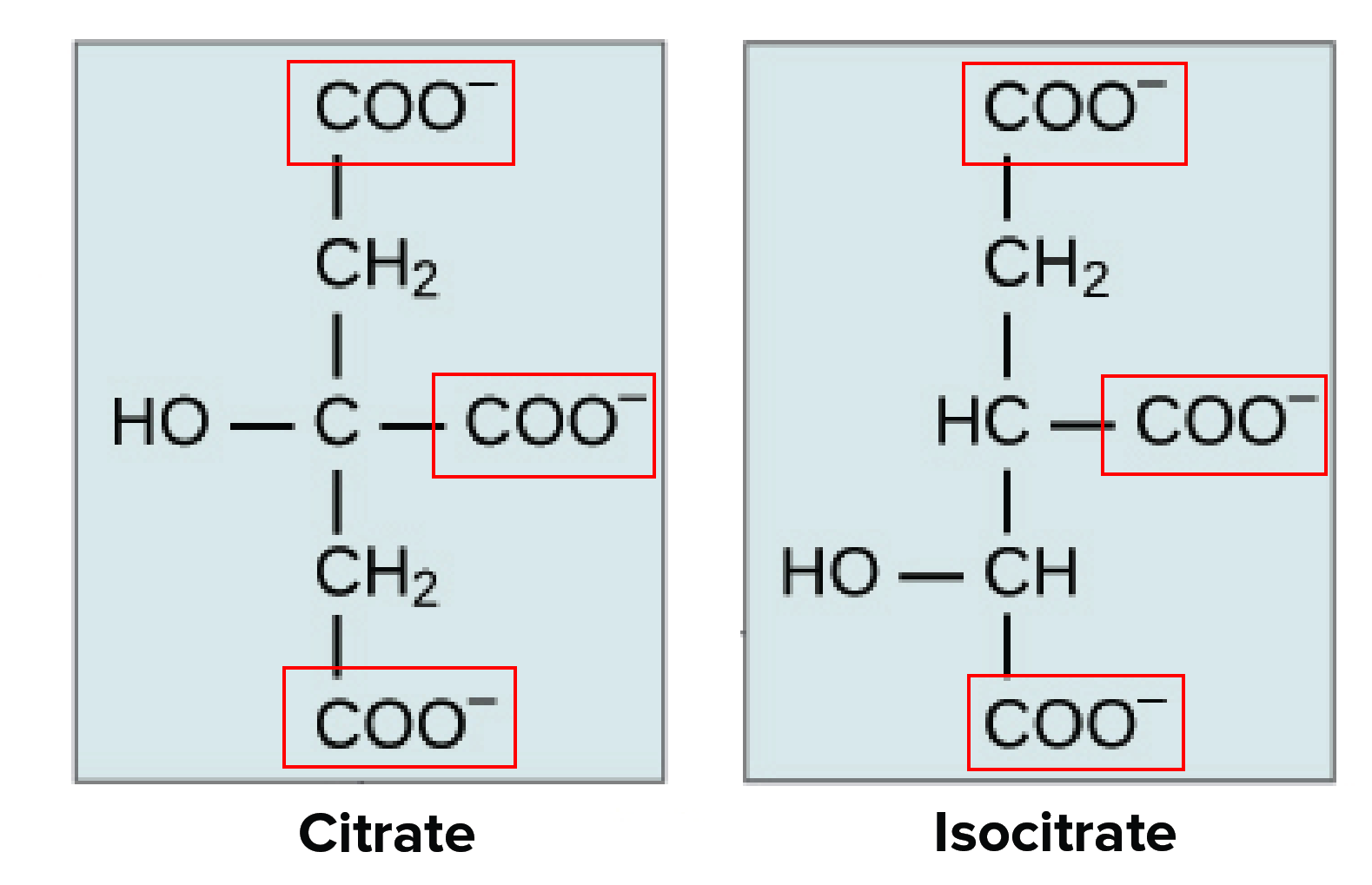
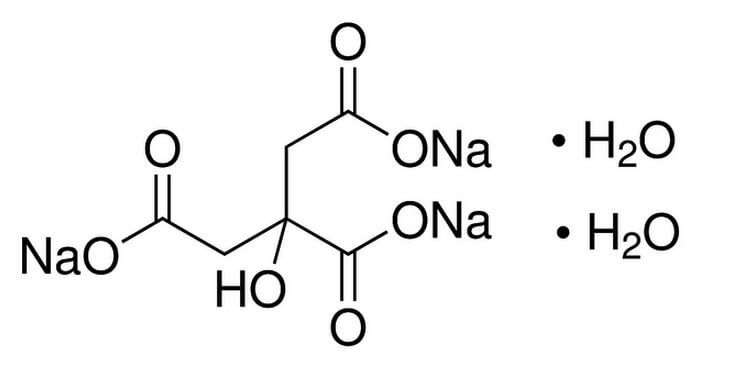
Safety Assessment of Citric Acid, Inorganic Citrate Salts, and Alkyl Citrate Esters as Used in Cosmetics - Monice M. Fiume, Bart A. Heldreth, Wilma F. Bergfeld, Donald V. Belsito, Ronald A. Hill,
Exogenous citrate can react with carbonic acid to form citric acid and... | Download Scientific Diagram

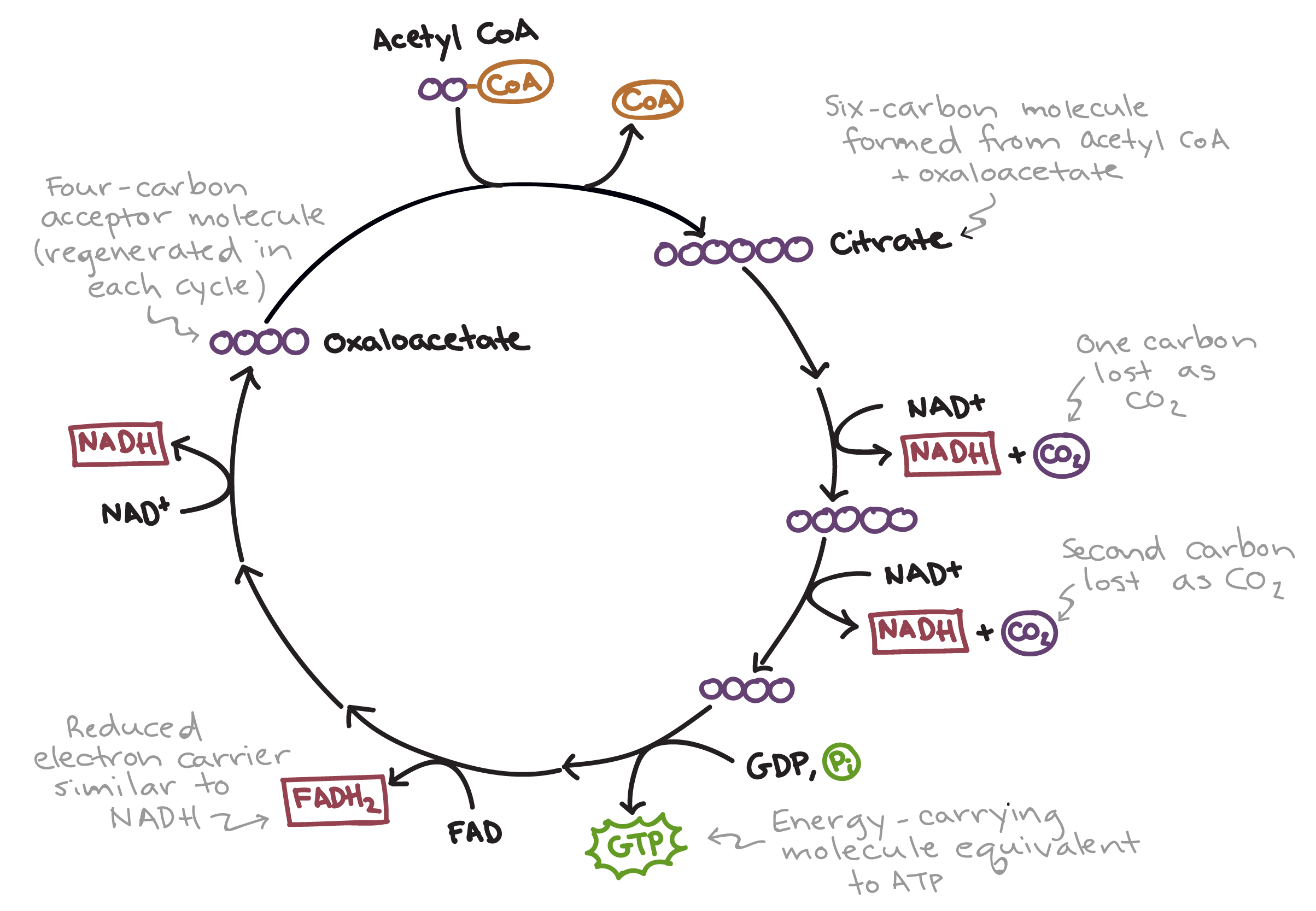
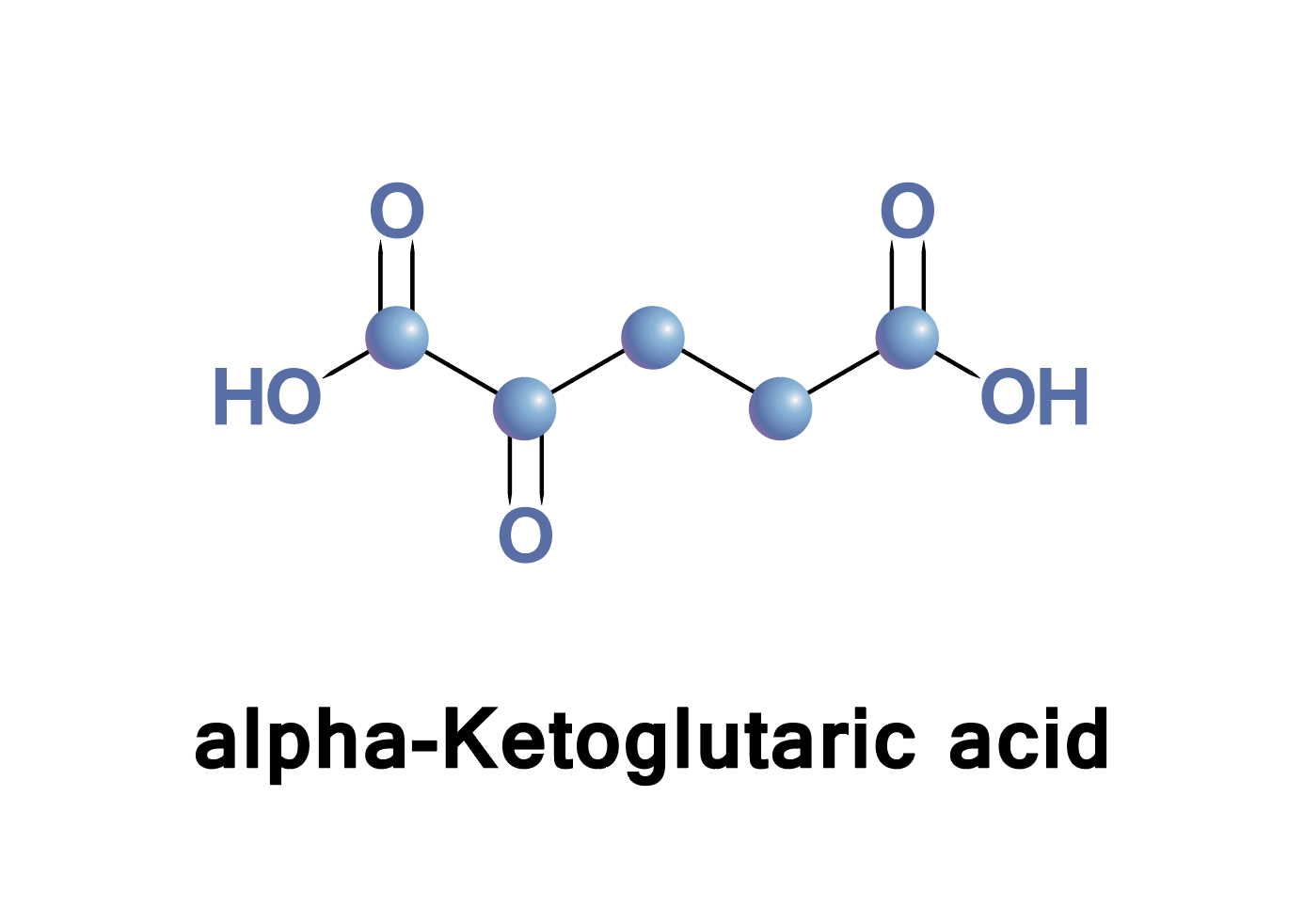
Tricarboxylic acid (TCA) cycle, the citric acid cycle or Krebs cycle. Citrate , cis-aconitate, isocitrate, α-ketoglutarate, succinate, fumarate, malate, oxaloacetate, oxalate, pyruvate, lactate, itaconate.

Safety Assessment of Citric Acid, Inorganic Citrate Salts, and Alkyl Citrate Esters as Used in Cosmetics - Monice M. Fiume, Bart A. Heldreth, Wilma F. Bergfeld, Donald V. Belsito, Ronald A. Hill,
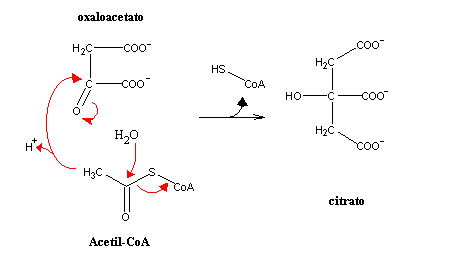
Non-Enzymatic Chemical Method for Dehydration of Citric acid Into Aconitic acid and Reaction Mechanisms