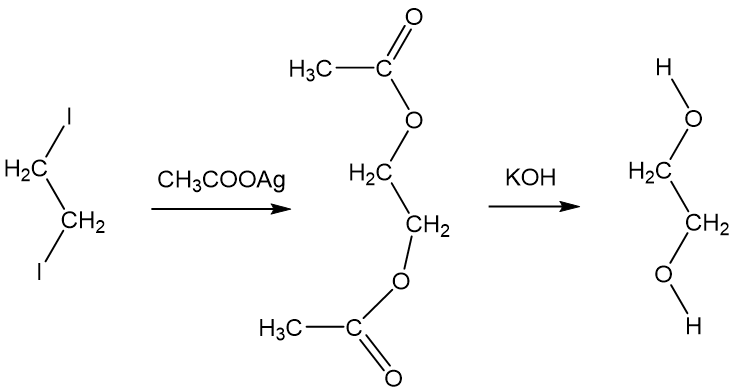
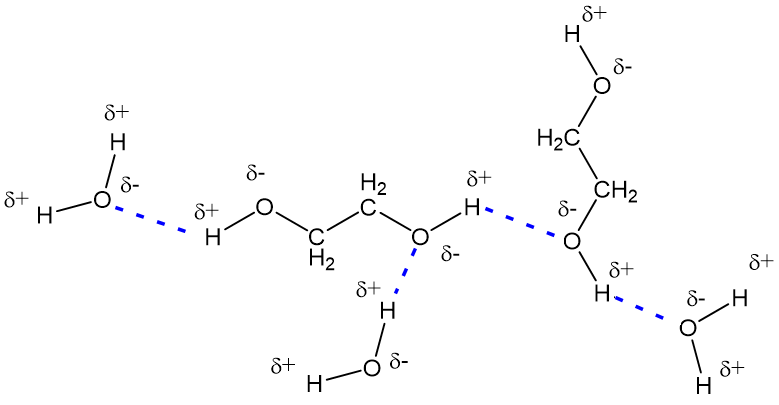
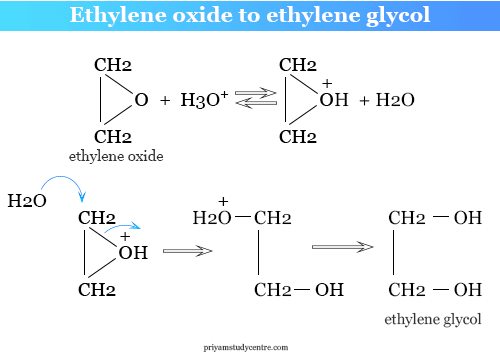
Enthalpies of reaction calculated from bond energies and from enthalpies of formation are often, but not always, close to each other. Ethylene glycol is produced by the catalytic oxidation of the ethy

Excess enthalpy of ethylene oxide + water at 293.15 K and 0.304 MPa:... | Download Scientific Diagram

Phase diagrams, densities and refractive indexes of poly(ethylene oxide) + organic salts + water aqueous two-phase systems: Effect of temperature, anion and molar mass - ScienceDirect
Flow diagram for a plant for hydrolysis of Ethylene Oxide (Arrieta,... | Download Scientific Diagram

Molecular Modeling and Simulation of Vapor–Liquid Equilibria of Ethylene Oxide, Ethylene Glycol, and Water as Well as their Binary Mixtures | Industrial & Engineering Chemistry Research

SciELO - Brasil - Water activity of aqueous solutions of ethylene oxide-propylene oxide block copolymers and maltodextrins Water activity of aqueous solutions of ethylene oxide-propylene oxide block copolymers and maltodextrins



.jpg)

.jpg)



![Poly(ethylene oxide) [MW 4,000,000] | Polysciences, Inc. Poly(ethylene oxide) [MW 4,000,000] | Polysciences, Inc.](https://www.polysciences.com/media/catalog/product/cache/c44c46ddedcd391a2ef31315a65fb4d0/0/4/04030.jpg)