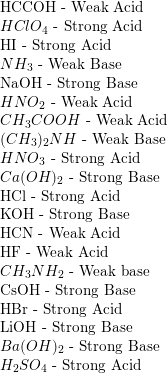
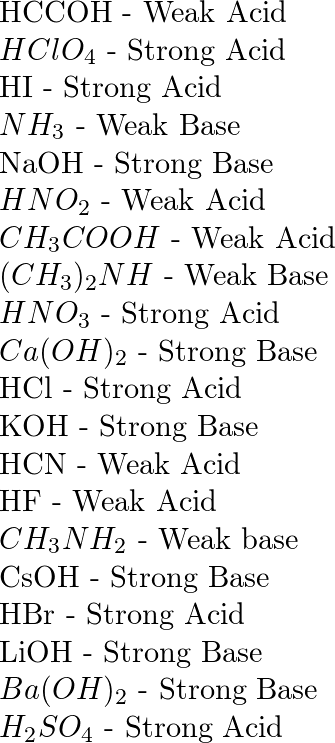SOLVED: 1. Calculate the pH of a 3.6 x 10^-4 M solution of HClO4, a strong acid, at 25ºC. 2. Rank the following acids in strength (from weakest to strongest). A HC2H3O2

SOLVED: Consider the substance HClO4. Part A How is HClO4 classified? neutral compound strong acid weak base basic salt weak acid acidic salt strong base Part B 17 1 point Item 17

HClO_(4),HNO_(3)` and `HCl` are all strong acids in aqueous solution.In glacial acetic acid medium, - YouTube
Perchloric acid (HClO4) molecule, chemical structure. Strong inorganic acid and oxidizer. Atoms are represented as spheres Stock Photo - Alamy