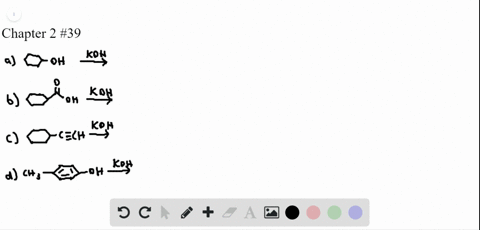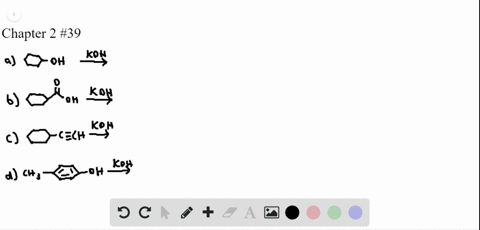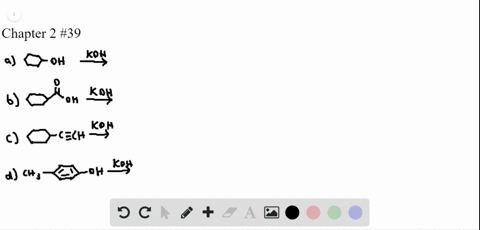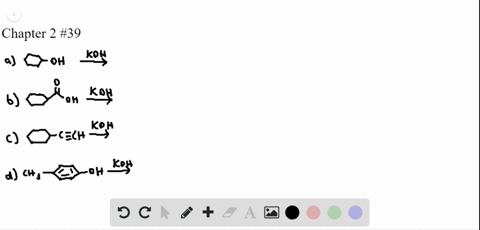
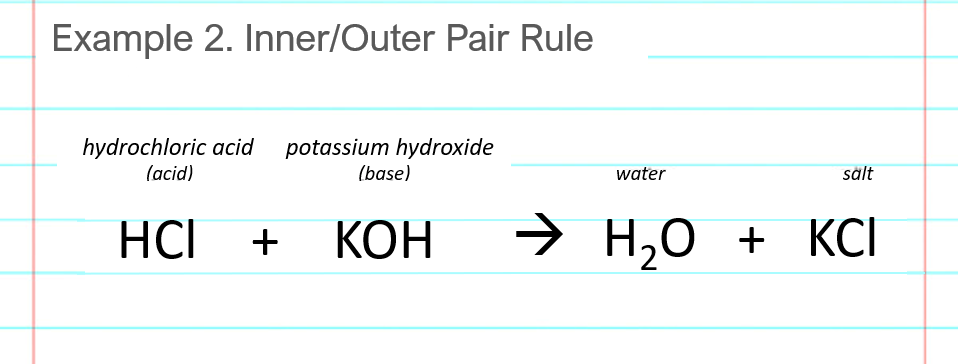
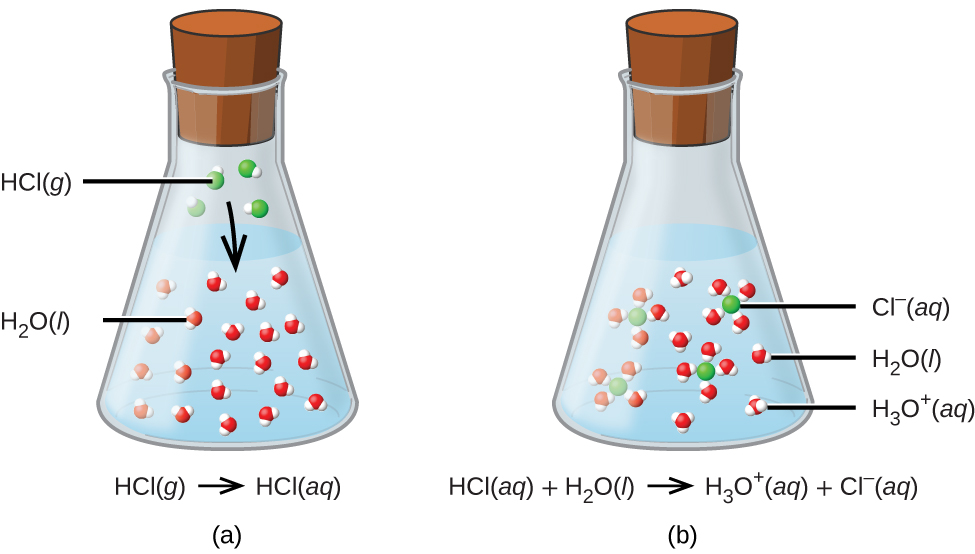
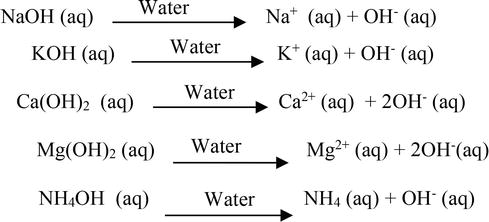
Chapter 3: Acid – Base Equilibria HCl + KOH KCl + H 2 O acid + base salt + water. - ppt download

SOLVED: List the conjugate acid or conjugate base for each chemical. a. The acid HF b. The base KOH c. The base NH3 d. The acid HNO3 e. The acid HCOOH f.