Boqu Nickel Sulfate Niso4 Nitric Acid Hno3 Oxalic Acid Ho2cco2h Water Quality In Industry Meter Analyzer - Buy Phosphoric Acid H3po4 Potassium Acetate Kch3co2 Potassium Bicarbonate Khco3 Water Analyzer Meter Probe,Potassium Biphthalate

TraceCERT Nickel Standard for ICP, 10,000 mg/L Ni in Nitric Acid, MilliporeSigma | Fisher Scientific
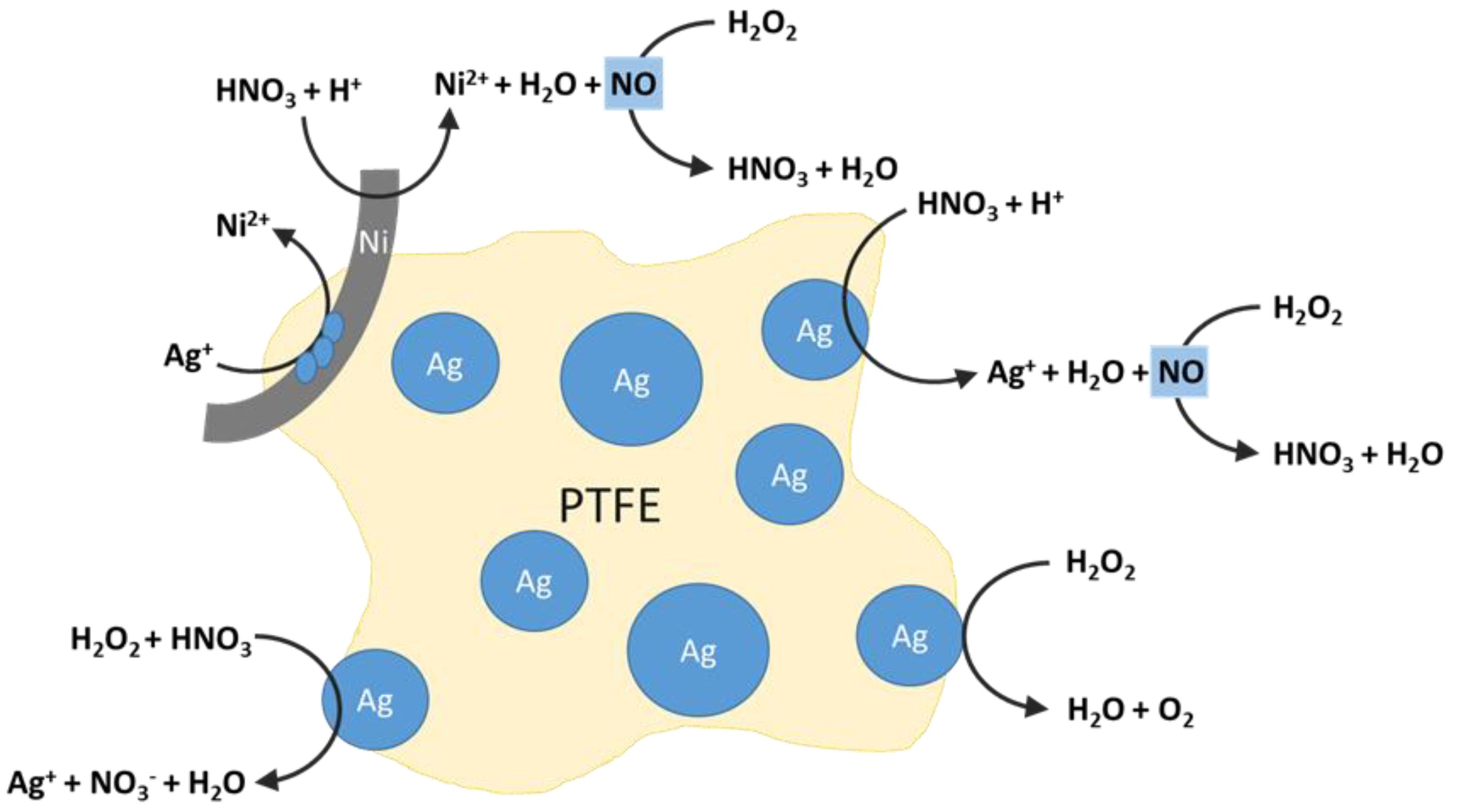
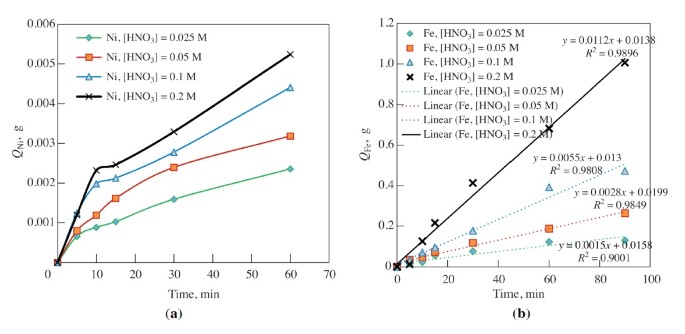
Metals | Free Full-Text | Kinetic Investigation of Silver Recycling by Leaching from Mechanical Pre-Treated Oxygen-Depolarized Cathodes Containing PTFE and Nickel
THE EQUILIBRIUM BETWEEN NITRIC OXIDE, NITROGEN PEROXIDE AND AQUEOUS SOLUTION OF NITRIC ACID. | Journal of the American Chemical Society
![PDF] Cobalt and Nickel Separation in Nitric Acid Solution by Solvent Extraction Using Cyanex 272 and Versatic 10 | Semantic Scholar PDF] Cobalt and Nickel Separation in Nitric Acid Solution by Solvent Extraction Using Cyanex 272 and Versatic 10 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ba83e1cf3818572a215179cf825a6ce6b007cc4b/6-Figure2-1.png)
PDF] Cobalt and Nickel Separation in Nitric Acid Solution by Solvent Extraction Using Cyanex 272 and Versatic 10 | Semantic Scholar

TraceCERT Nickel Standard for ICP, 10,000 mg/L Ni in Nitric Acid, MilliporeSigma | Fisher Scientific

Optimization of process parameters for the selective leaching of copper, nickel and isolation of gold from obsolete mobile phone PCBs - ScienceDirect

Selective pressure leaching of Fe (II)-rich limonitic laterite ores from Indonesia using nitric acid - ScienceDirect