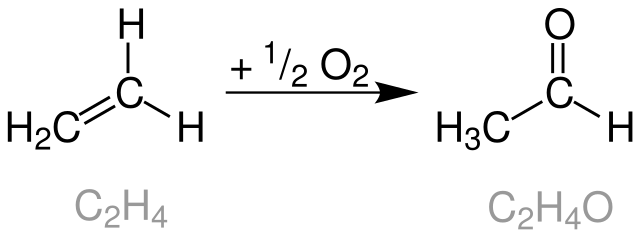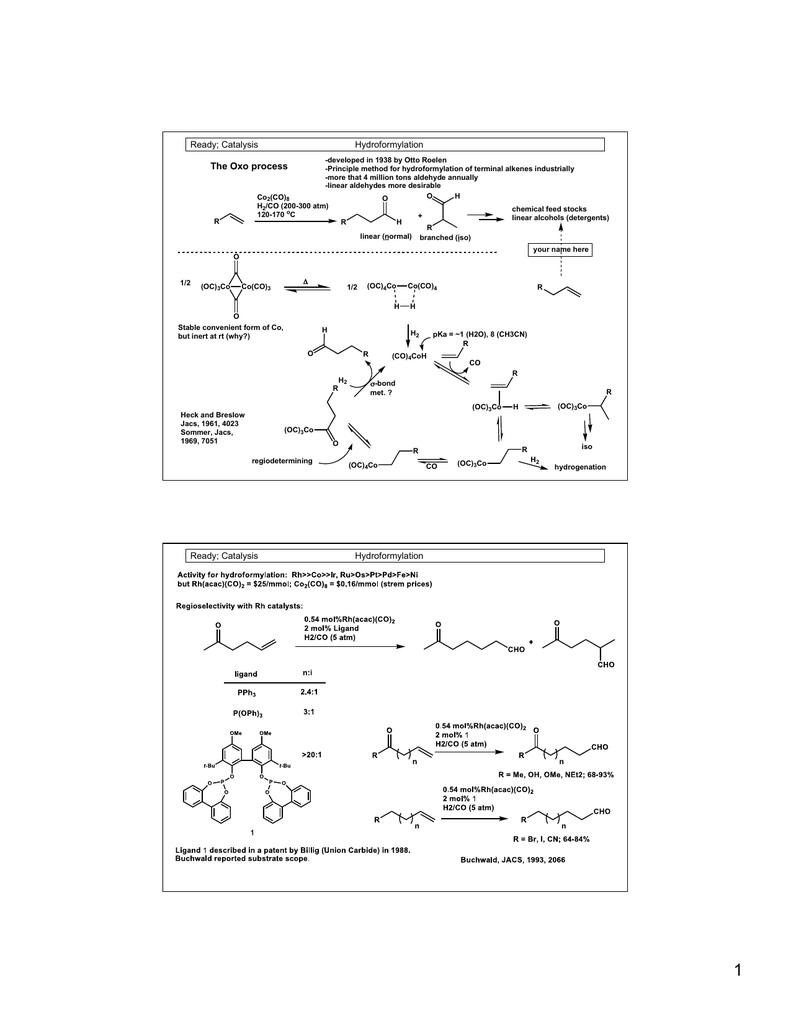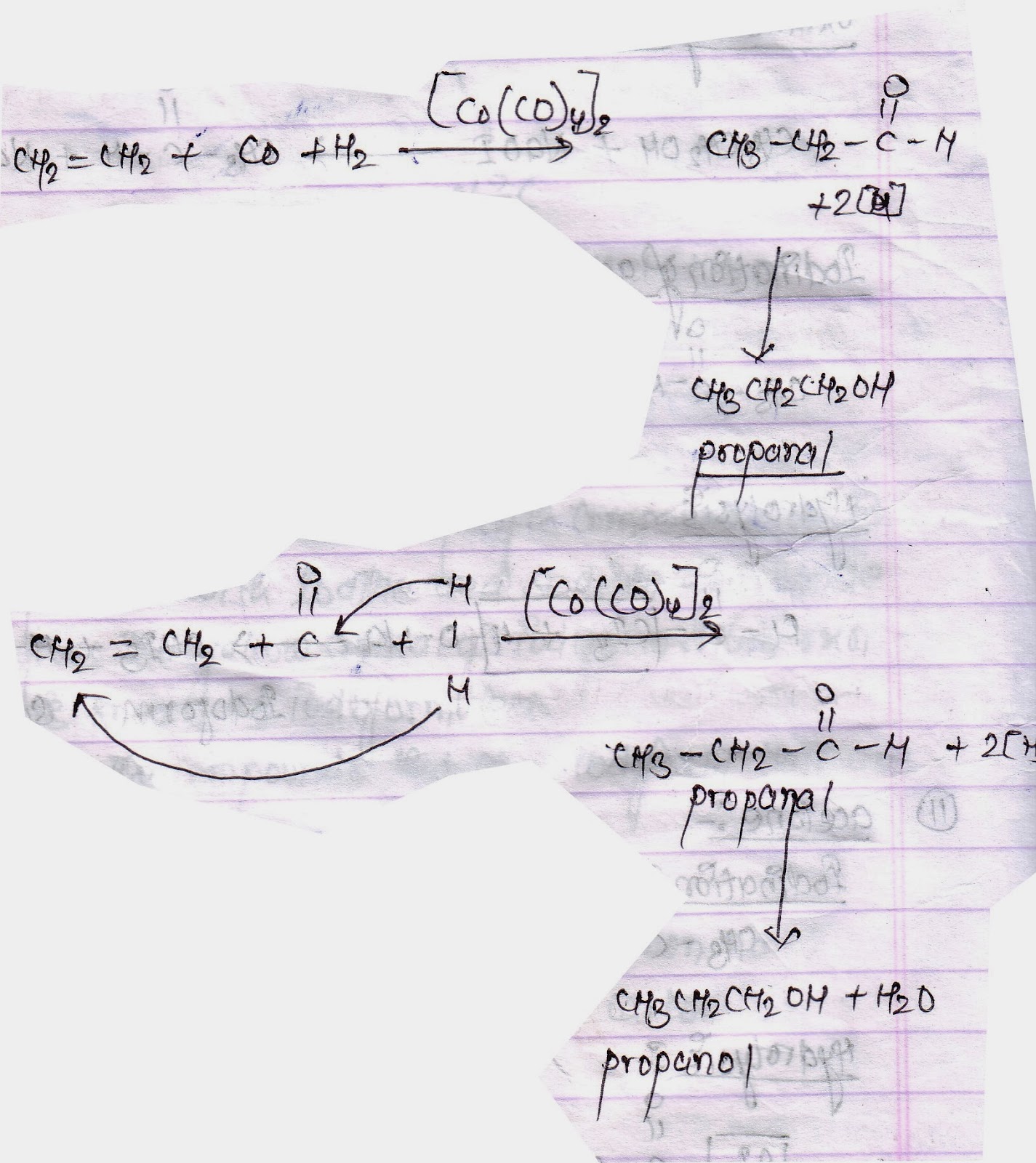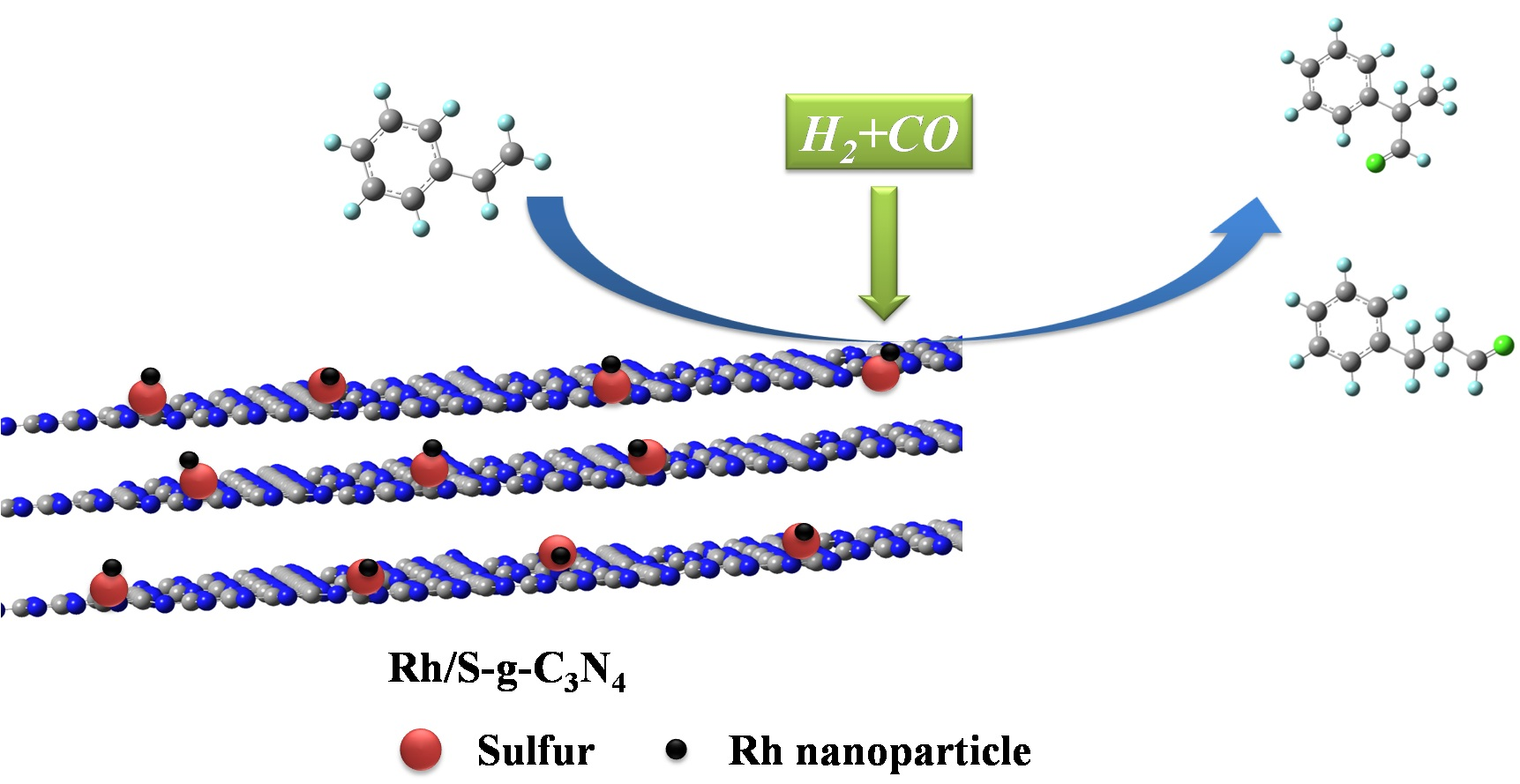
Catalysts | Free Full-Text | Rh Particles Supported on Sulfated g-C3N4: A Highly Efficient and Recyclable Heterogeneous Catalyst for Alkene Hydroformylation
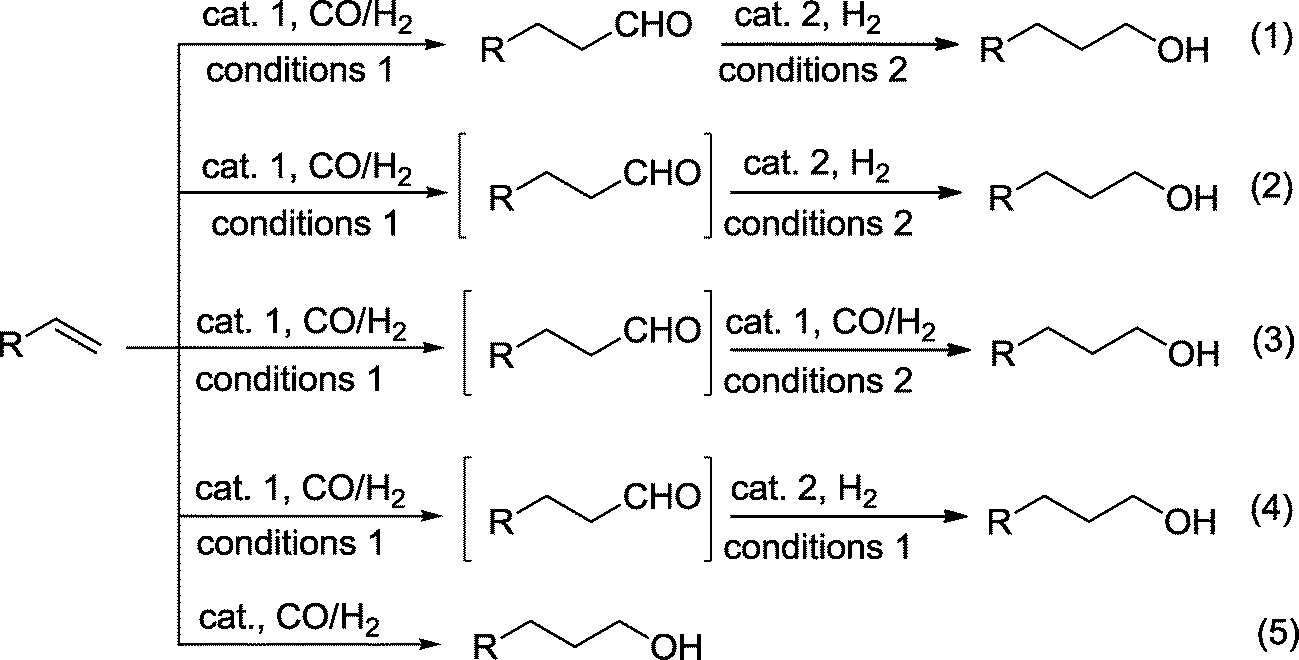
Production of alcohols via hydroformylation - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C4CY01131G
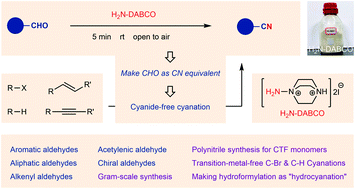
Momentary click nitrile synthesis enabled by an aminoazanium reagent - Organic Chemistry Frontiers (RSC Publishing)

A Strategy for Amide to β-Oxo Ester Transformation via N-Alkenoxypyridinium Salts as the Activator and H2O as the Nucleophile | Organic Letters
Amine-resin supported rhodium-cobalt bimetallic clusters as hydroformylation catalysts, their preparation and oxo process utilising such a catalyst - Patent 0014225

Synthesis of a boron-containing amidoxime reagent and its application to synthesize functionalized oxadiazole and quinazolinone derivatives - ScienceDirect

Exchange-enhanced reactivity in bond activation by metal–oxo enzymes and synthetic reagents | Nature Chemistry

![Hydroformylation of Olefins by OXO PROCESS | | [Co2(CO)8] Catalyst - YouTube Hydroformylation of Olefins by OXO PROCESS | | [Co2(CO)8] Catalyst - YouTube](https://i.ytimg.com/vi/zIiOix_Y7tc/sddefault.jpg)