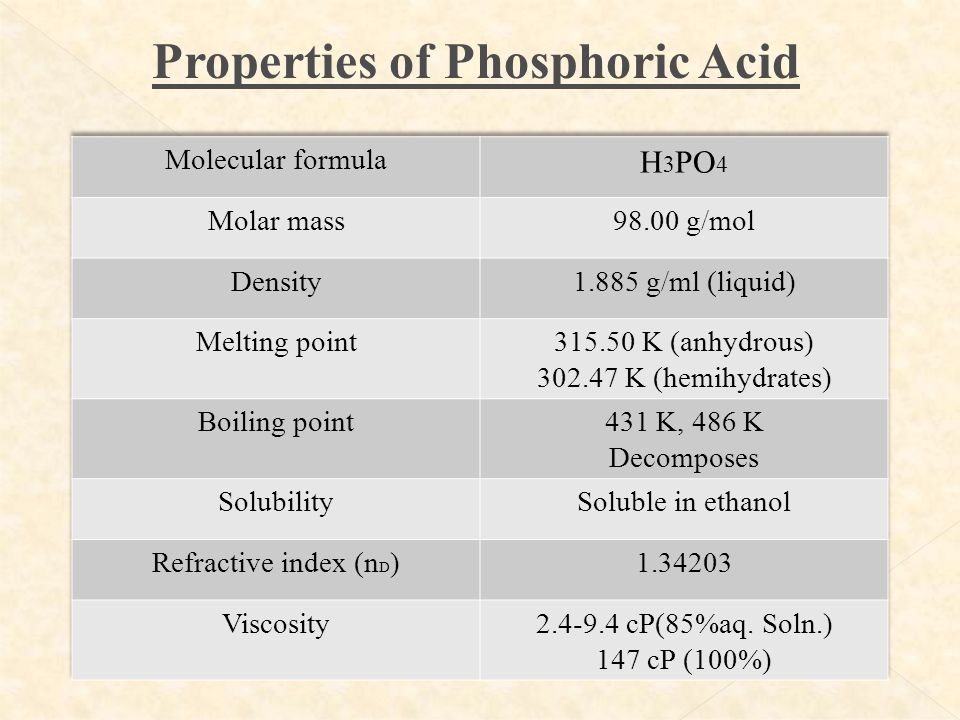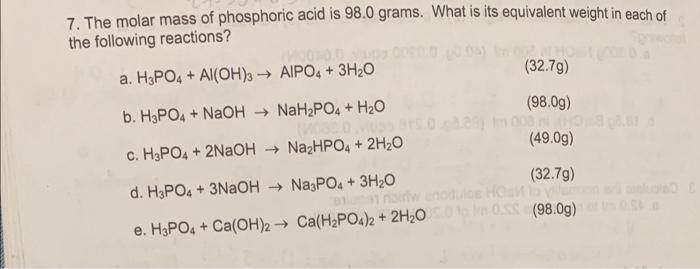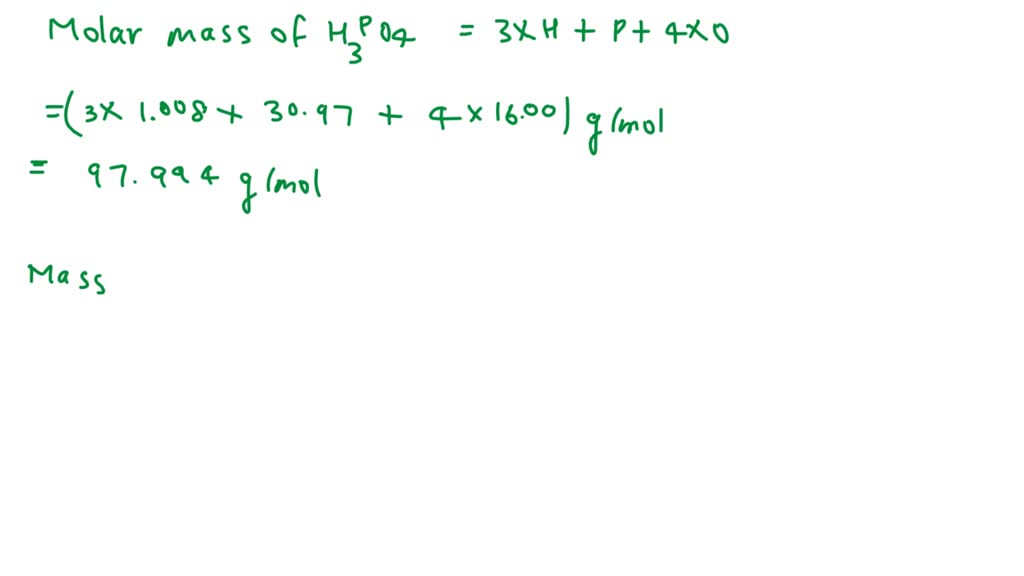
SOLVED: What is the percent of phosphorus by mass in phosphoric acid, H3PO4? Use 1.008 g/mol for the molar mass of hydrogen, 16.00 g/mol for the molar mass of oxygen, and 30.97

SOLVED: The morality of a 30.5% by mass aqueous solution of phosphoric acid H3PO4 is. molar mass of H3PO4 = 97.99g/mol

SOLVED: The commercial production of phosphoric acid, HzPOA can be represented by the equation 1580 g 296 g 317 g 1030 g 296 g Caz(PO4)2 3Si02 SC + 502 3HzO 3CaSiOg SCO2 +

The equivalent weight of phosphoric acid `(H_(3)PO_(4))` in the reaction `NaOH+H_(3)PO_(4) rarr NaH_ - YouTube
![Orthophosphoric Acid (H3PO4) [Phosphoric Acid] - Structure, Formula, Synthesis, Properties, Uses and FAQs of Orthophosphoric acid. Orthophosphoric Acid (H3PO4) [Phosphoric Acid] - Structure, Formula, Synthesis, Properties, Uses and FAQs of Orthophosphoric acid.](https://cdn1.byjus.com/wp-content/uploads/2018/11/phosphoric-acid-structure.png)
Orthophosphoric Acid (H3PO4) [Phosphoric Acid] - Structure, Formula, Synthesis, Properties, Uses and FAQs of Orthophosphoric acid.
Q. The equivalent mass of H3PO4 (Molecular weight = 98 g/mol) and Na2HPO4 (Molecular weight = 142 g/mol) in the reaction are respectively : H3PO4 + 2NaOH → Na2HPO4 + 2H2O (1) 49, 142 (2) 49, 71 (3) 98, 71 (4) 98, 142

OneClass: Calculate molality of a 35.4% (by mass) aqueous solution of phosphoric acid (H3PO4). The mo...