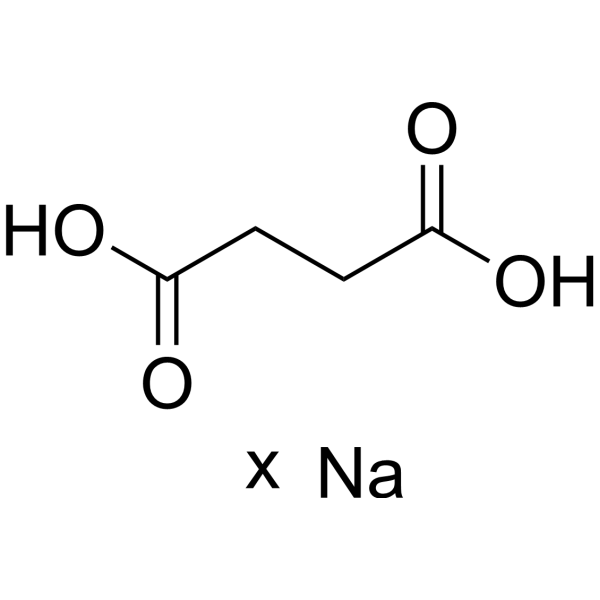
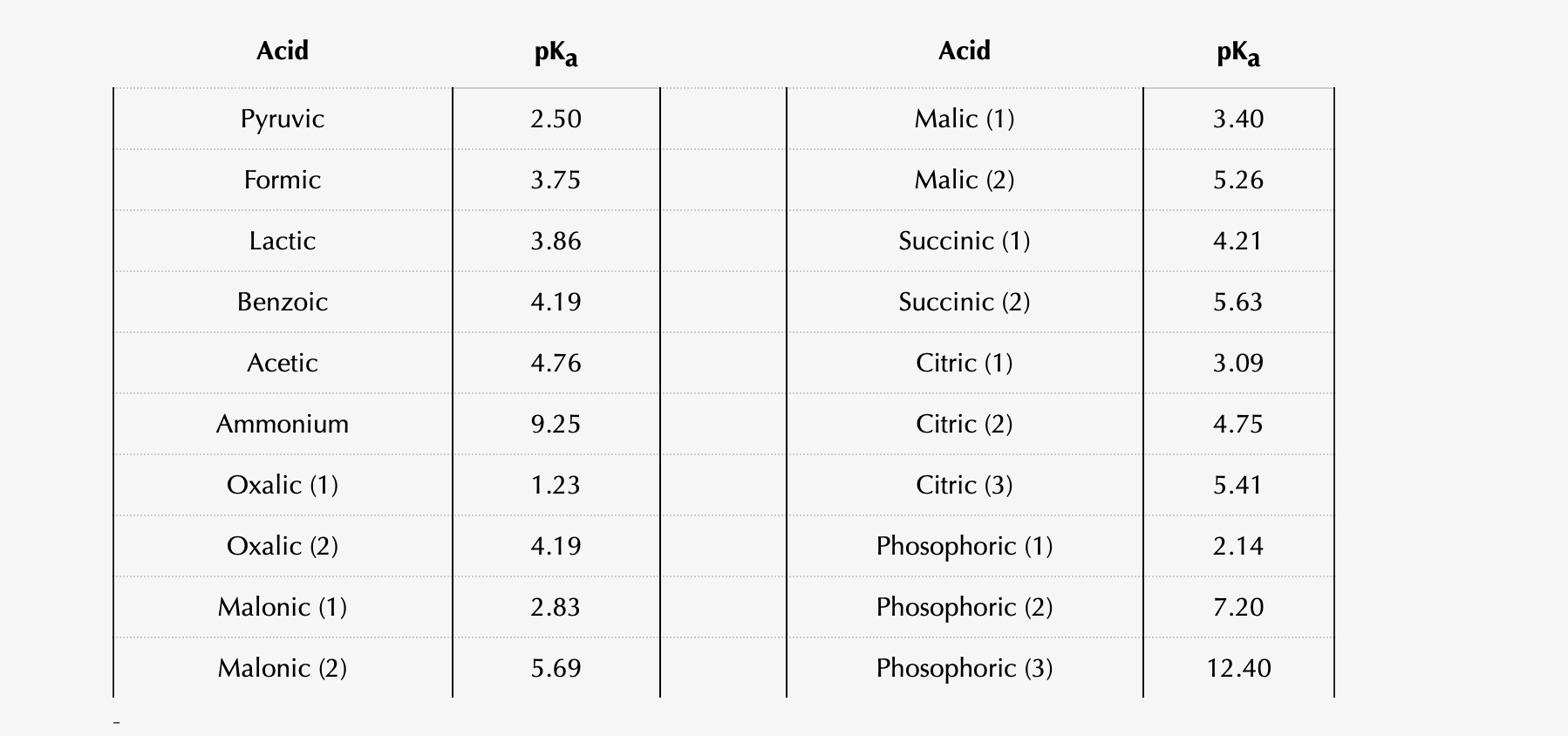
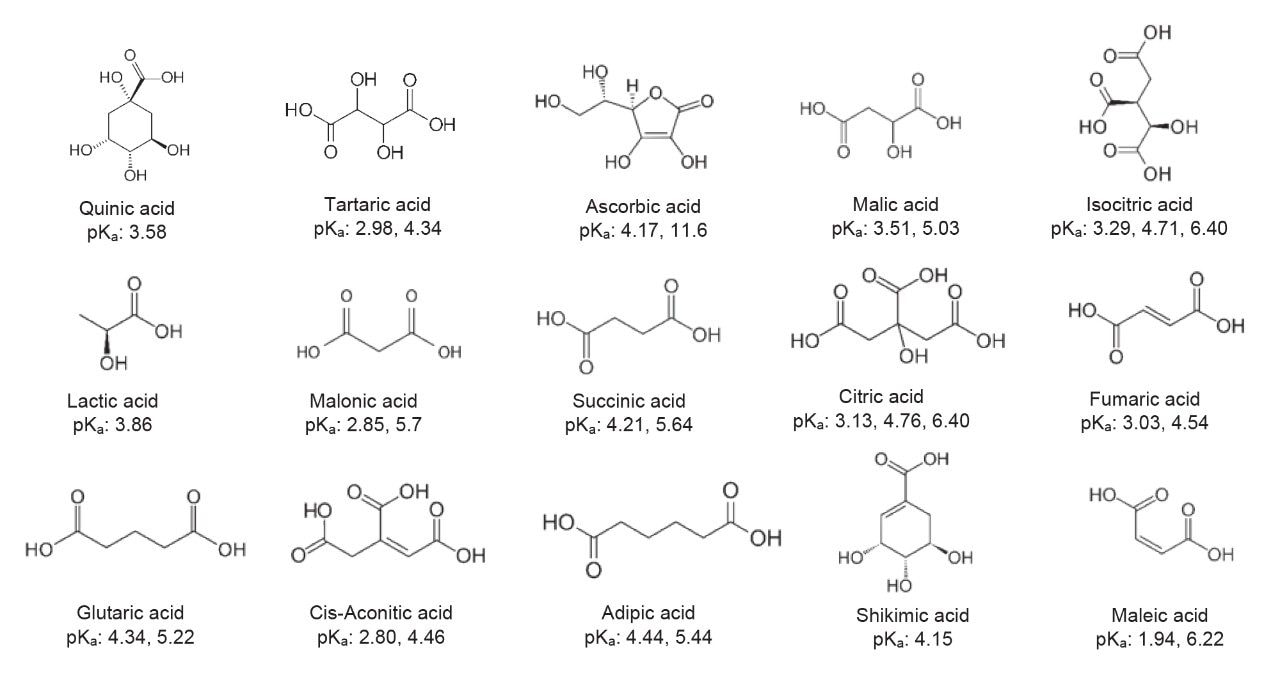
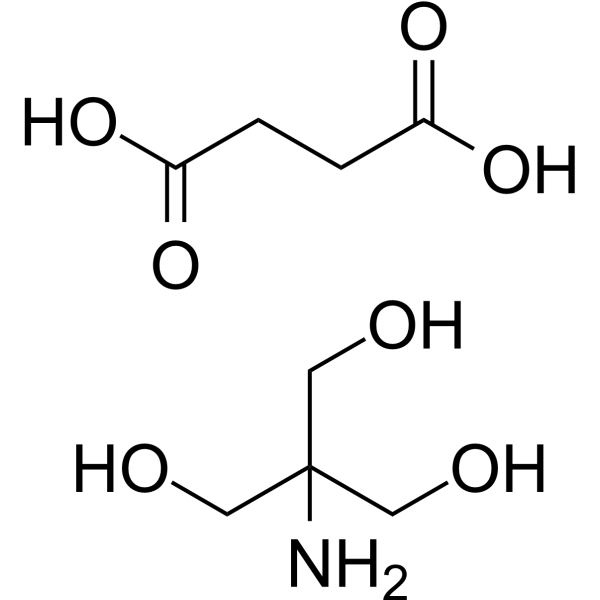
A Reliable and Efficient First Principles-Based Method for Predicting pKa Values. 2. Organic Acids | The Journal of Physical Chemistry A

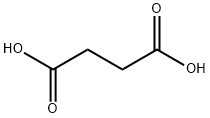
Within the buffer range of succinic acid (pKa 1 = 4.16, pKa 2 = 5.61) H... | Download Scientific Diagram

OneClass: How many grams of dipotassium succinate trihydrate (KpC4H4O4. 3H20, MW = 248.32 g/mol) must...
Industrial Systems Biology of Saccharomyces cerevisiae Enables Novel Succinic Acid Cell Factory | PLOS ONE
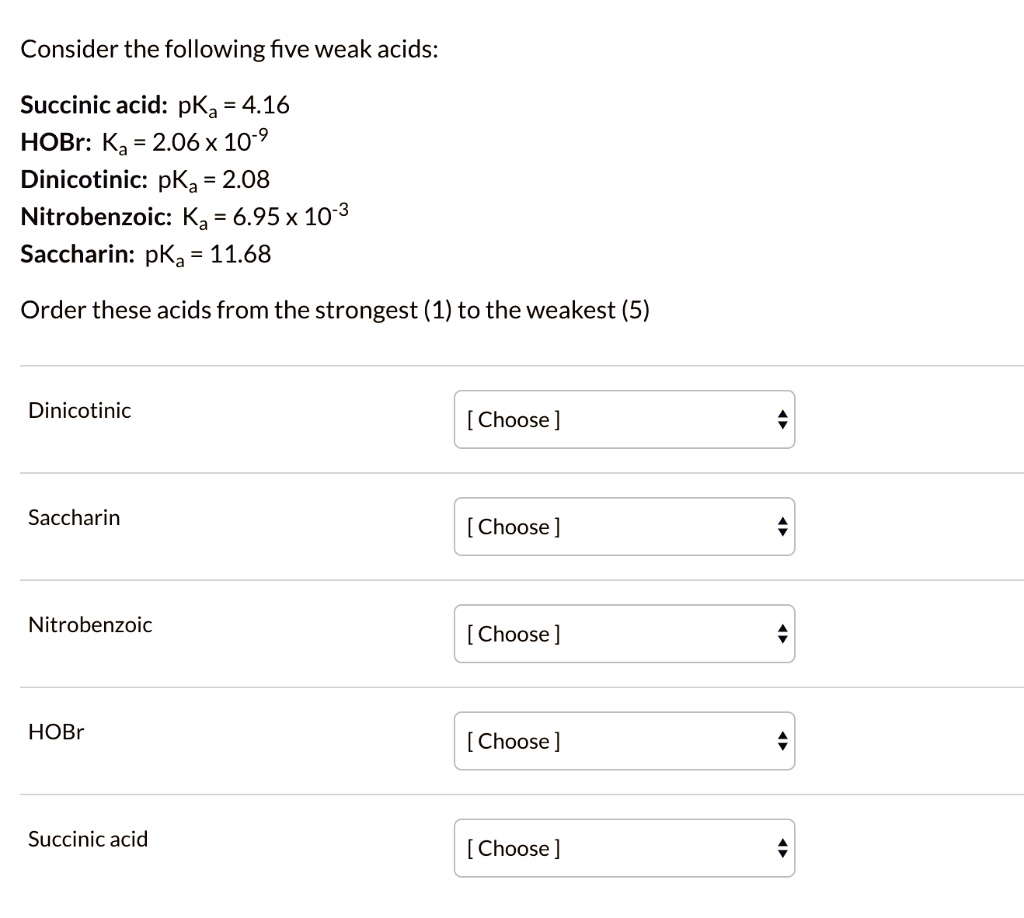
SOLVED: Consider the following five weak acids: Succinic acid: pKa 4.16 HOBr: Ka = 2.06X 10-9 Dinicotinic: pKa 2.08 Nitrobenzoic: Ka 6.95X 10-3 Saccharin: pKa 11.68 Order these acids from the strongest (
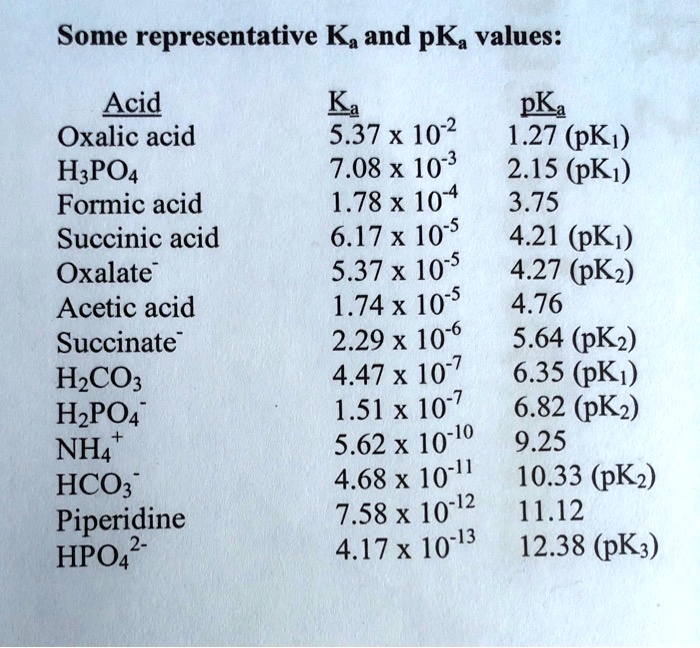
SOLVED: Some representative Ka and pKa values: Acid Oxalic acid HzPOa Formic acid Succinic acid Oxalate Acetic acid Succinate HzCOz HzPOA NH4' HCO; Piperidine HPOA2 Ka pKa 5.37 x 10-2 1.27 (pK,)