Recent attempts to change the periodic table | Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences

Notes 4 ions - CHEM 1a Energy Levels in Many-Electron Atoms The following figure shows the electron - Studocu

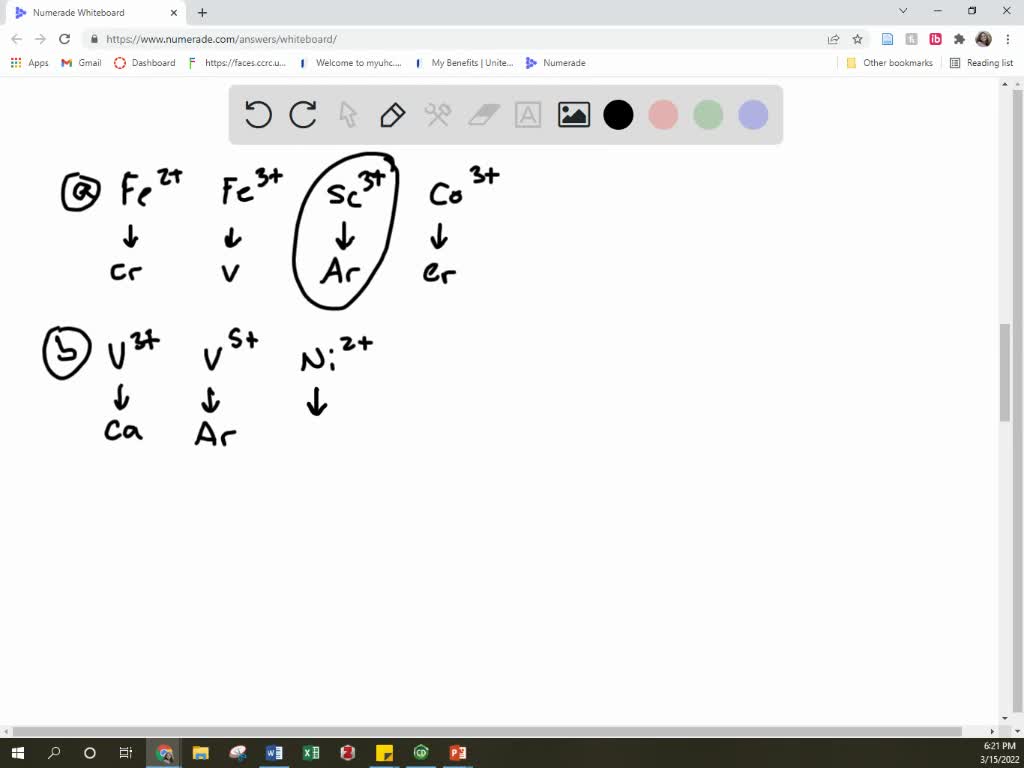
OneClass: Predict the ground-state electron configuration of the following ions. Write your answers i...

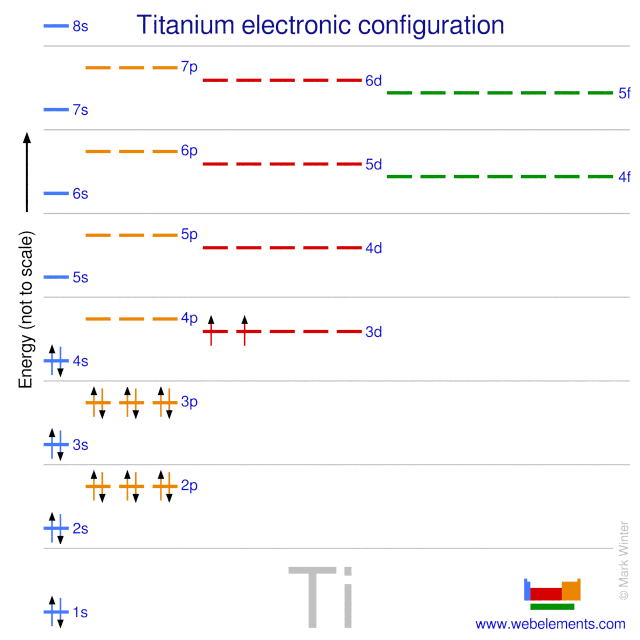
Electronic states of neutral and cationic bis(benzene) titanium and vanadium sandwich complexes studied by pulsed field ionization electron spectroscopy: The Journal of Chemical Physics: Vol 127, No 11

Write the condensed ground-state electron configuration for Cd2+. Is it paramagnetic or diamagnetic? | Homework.Study.com

SOLVED: Which of the following ions have noble gas electron configurations? (Select all that apply: (a) Y3+, Mn4+, Fe 3+ V2+ Y3+ Mn4+ Fe3+ vz+ (b) T1+ , Ti3+, 02- TI+ TI3+
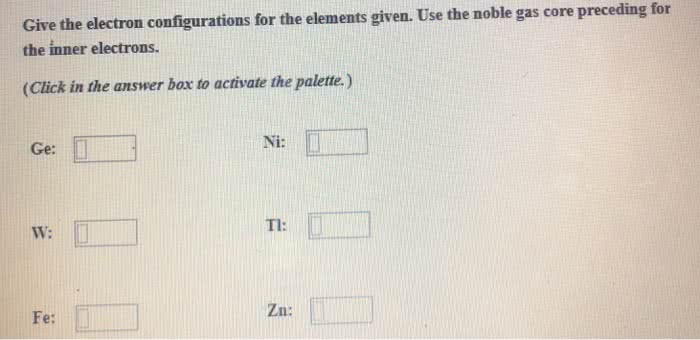
OneClass: Give the electron configurations for the elements given. Use the noble gas core preceding f...